

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50304324 ((1R,9R)-1-{[1-(3,5-Dimethoxy-phenyl)-methylidene]-...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese Academy of Sciences Curated by ChEMBL | Assay Description Inhibition of Torpedo california AChE by Ellman's method | Bioorg Med Chem 17: 6937-41 (2009) Article DOI: 10.1016/j.bmc.2009.08.017 BindingDB Entry DOI: 10.7270/Q2J966GQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9022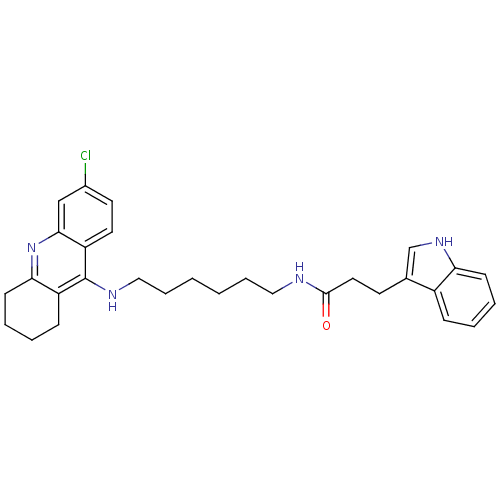 (CHEMBL225567 | Indole-Tacrine Heterodimer 5 | N-[5...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50304322 ((1R,9R)-13-Eth-(E)-ylidene-1-{[1-(4-methoxy-phenyl...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0246 | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese Academy of Sciences Curated by ChEMBL | Assay Description Inhibition of Torpedo california AChE by Ellman's method | Bioorg Med Chem 17: 6937-41 (2009) Article DOI: 10.1016/j.bmc.2009.08.017 BindingDB Entry DOI: 10.7270/Q2J966GQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50304326 (4-{[13-Ethylidene-11-methyl-5-oxo-6-aza-tricyclo[7...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0412 | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese Academy of Sciences Curated by ChEMBL | Assay Description Inhibition of Torpedo california AChE by Ellman's method | Bioorg Med Chem 17: 6937-41 (2009) Article DOI: 10.1016/j.bmc.2009.08.017 BindingDB Entry DOI: 10.7270/Q2J966GQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50304325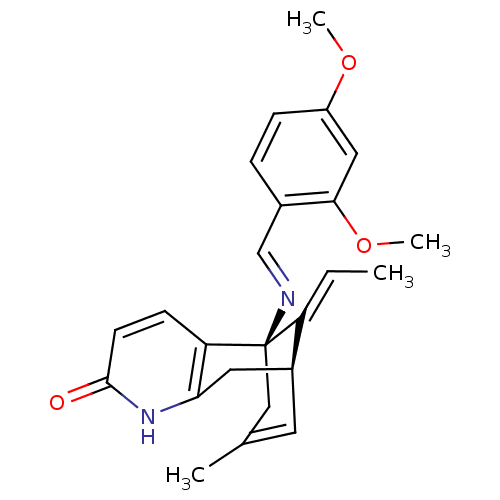 (1-{[1-(2,4-Dimethoxy-phenyl)-methylidene]-amino}-1...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0446 | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese Academy of Sciences Curated by ChEMBL | Assay Description Inhibition of Torpedo california AChE by Ellman's method | Bioorg Med Chem 17: 6937-41 (2009) Article DOI: 10.1016/j.bmc.2009.08.017 BindingDB Entry DOI: 10.7270/Q2J966GQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9023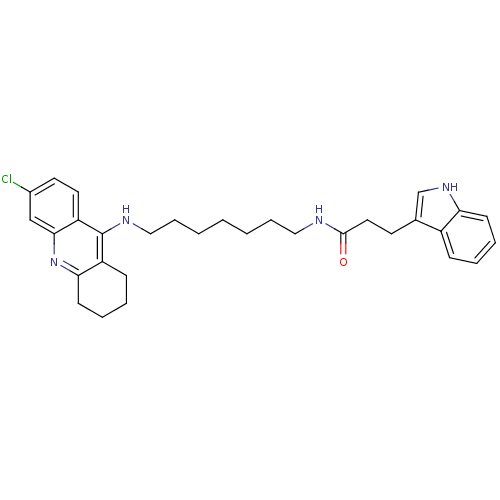 (CHEMBL225198 | Indole-Tacrine Heterodimer 6 | N-[7...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM9054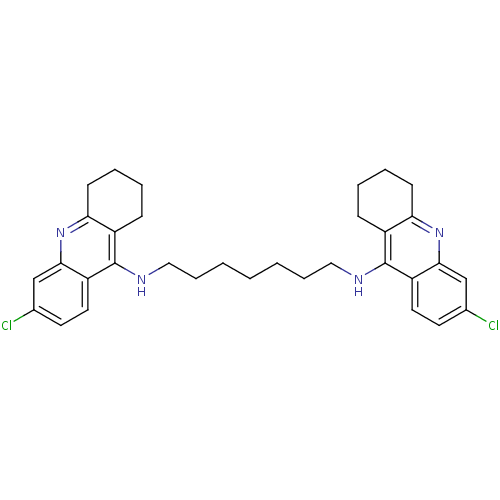 (6-chloro-N-{7-[(6-chloro-1,2,3,4-tetrahydroacridin...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | 7.4 | 37 |
National Defense Medical Center | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. Enzyme activity was determined by measuring the absorbance at... | J Med Chem 45: 2277-82 (2002) Article DOI: 10.1021/jm010308g BindingDB Entry DOI: 10.7270/Q2KW5D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50235322 (CHEMBL4066593) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences Curated by ChEMBL | Assay Description Inhibition of rat cortex AchE using acetylthiocholine iodide as substrate by Ellman's method | Eur J Med Chem 128: 332-345 (2017) Article DOI: 10.1016/j.ejmech.2016.10.060 BindingDB Entry DOI: 10.7270/Q2HM5BQX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271142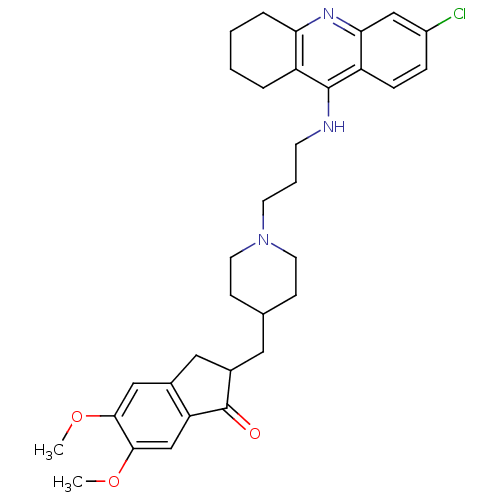 (6-Chloro-9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50262988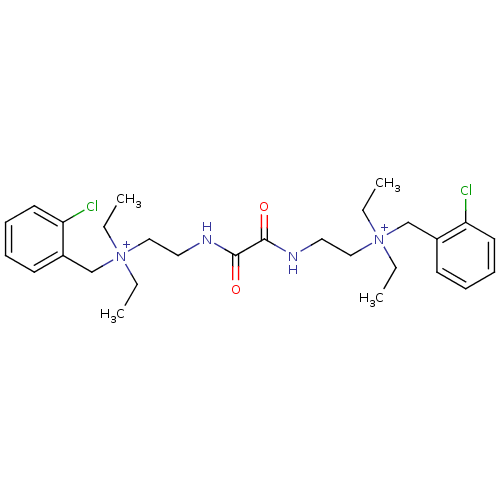 (CHEMBL1200541 | N-(2-chlorobenzyl)-2-(2-(2-((2-chl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Bari Curated by ChEMBL | Assay Description Inhibition of bovine AChE by Ellman's method | Bioorg Med Chem 16: 7450-6 (2008) Article DOI: 10.1016/j.bmc.2008.06.022 BindingDB Entry DOI: 10.7270/Q2NC611F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50304330 ((1R,9R)-1-[3-(4-Dimethylamino-phenyl)-prop-2-enyli...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.121 | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese Academy of Sciences Curated by ChEMBL | Assay Description Inhibition of Torpedo california AChE by Ellman's method | Bioorg Med Chem 17: 6937-41 (2009) Article DOI: 10.1016/j.bmc.2009.08.017 BindingDB Entry DOI: 10.7270/Q2J966GQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50262879 (3-hydroxy-5-(4-(3-hydroxy-5-(trimethylammonio)phen...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Bari Curated by ChEMBL | Assay Description Inhibition of bovine AChE by Ellman's method | Bioorg Med Chem 16: 7450-6 (2008) Article DOI: 10.1016/j.bmc.2008.06.022 BindingDB Entry DOI: 10.7270/Q2NC611F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9009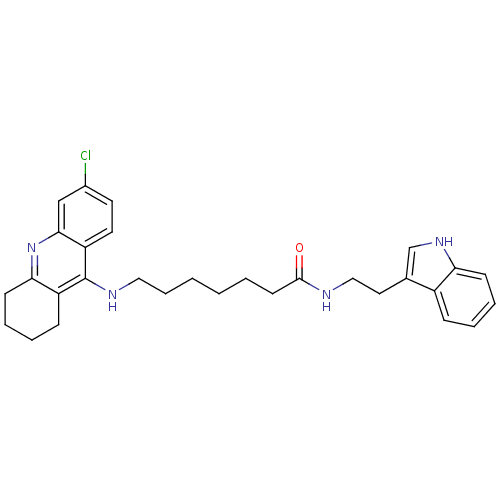 (7-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Instituto de Quimica Medica (CSIC) | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 412 nm were recorded for 5 min with ... | J Med Chem 49: 459-62 (2006) Article DOI: 10.1021/jm050746d BindingDB Entry DOI: 10.7270/Q2VD6WN2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9009 (7-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences Curated by ChEMBL | Assay Description Inhibitory activity was determined against bovine liver beta galactosidase | Eur J Med Chem 128: 332-345 (2017) Article DOI: 10.1016/j.ejmech.2016.10.060 BindingDB Entry DOI: 10.7270/Q2HM5BQX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9035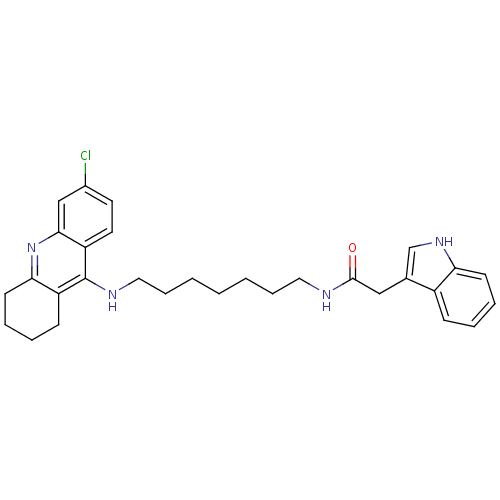 (Indole-Tacrine Heterodimer 18 | N-[7-(6-Chloro-1,2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM8963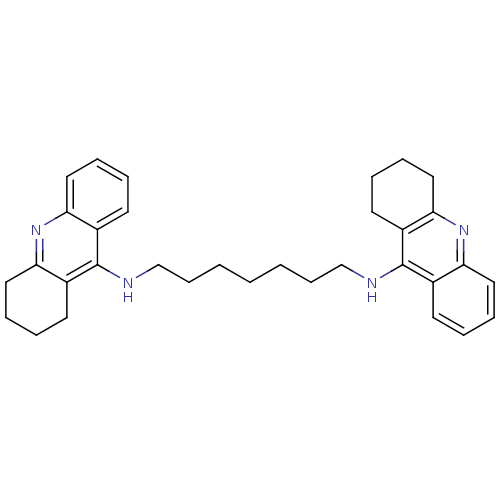 (CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | 7.4 | 37 |
National Defense Medical Center | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. Enzyme activity was determined by measuring the absorbance at... | J Med Chem 45: 2277-82 (2002) Article DOI: 10.1021/jm010308g BindingDB Entry DOI: 10.7270/Q2KW5D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50235323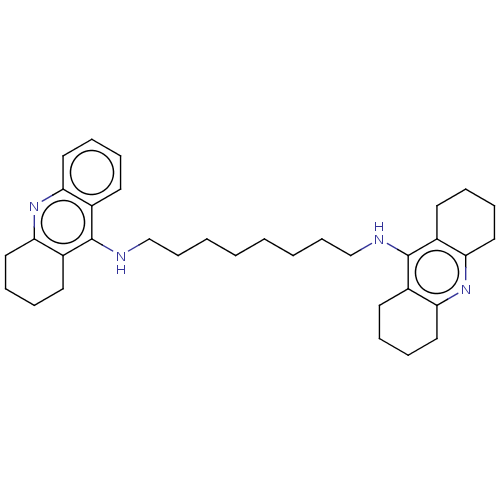 (CHEMBL4080023) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences Curated by ChEMBL | Assay Description Inhibition of rat cortex AchE using acetylthiocholine iodide as substrate by Ellman's method | Eur J Med Chem 128: 332-345 (2017) Article DOI: 10.1016/j.ejmech.2016.10.060 BindingDB Entry DOI: 10.7270/Q2HM5BQX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50369748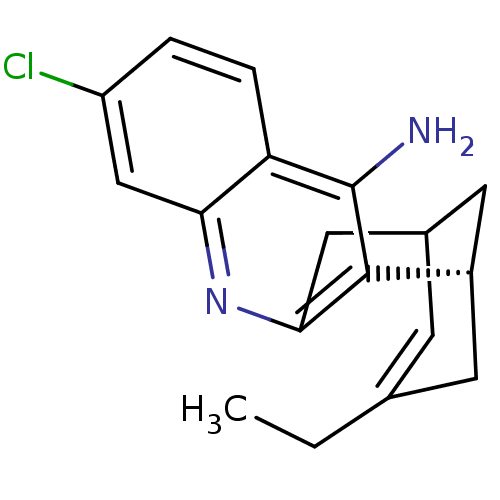 (CHEMBL208599) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50369748 (CHEMBL208599) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271140 (9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)methyl]pi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM9410 (N-[2-(1-benzylpiperidin-4-yl)ethyl]-N-ethyl-4-(phe...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Laboratories | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The amount of a yellow substance formed after incubation was d... | J Med Chem 33: 1880-7 (1990) Article DOI: 10.1021/jm00169a008 BindingDB Entry DOI: 10.7270/Q20Z71H2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM9055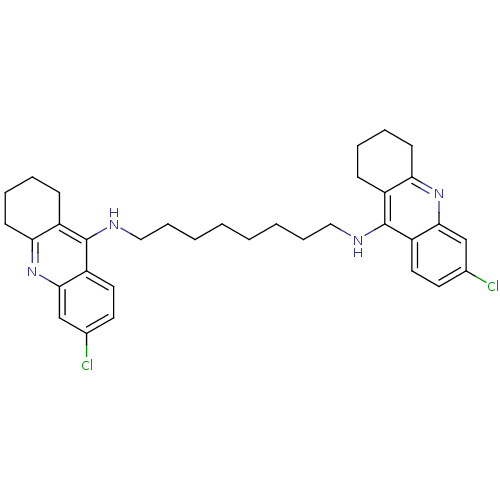 (6-chloro-N-{8-[(6-chloro-1,2,3,4-tetrahydroacridin...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | 7.4 | 37 |
National Defense Medical Center | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. Enzyme activity was determined by measuring the absorbance at... | J Med Chem 45: 2277-82 (2002) Article DOI: 10.1021/jm010308g BindingDB Entry DOI: 10.7270/Q2KW5D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9037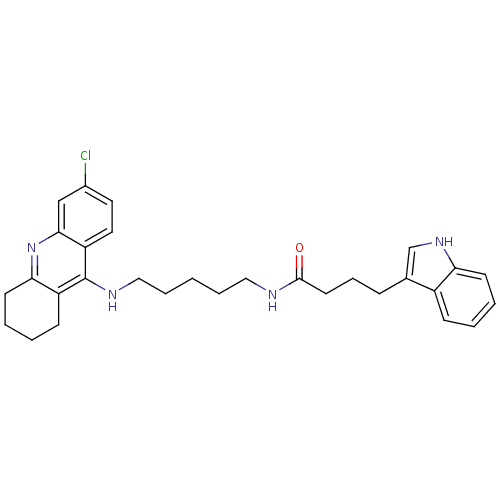 (Indole-Tacrine Heterodimer 20 | N-[5-(6-Chloro-1,2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50235324 (CHEMBL4104258) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences Curated by ChEMBL | Assay Description Inhibition of rat cortex AchE using acetylthiocholine iodide as substrate by Ellman's method | Eur J Med Chem 128: 332-345 (2017) Article DOI: 10.1016/j.ejmech.2016.10.060 BindingDB Entry DOI: 10.7270/Q2HM5BQX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM9410 (N-[2-(1-benzylpiperidin-4-yl)ethyl]-N-ethyl-4-(phe...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.301 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Missouri-St. Louis Curated by ChEMBL | Assay Description Inhibition against Acetylcholinesterase (AChE) | J Med Chem 39: 380-7 (1996) Article DOI: 10.1021/jm950704x BindingDB Entry DOI: 10.7270/Q25D8T1Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50037158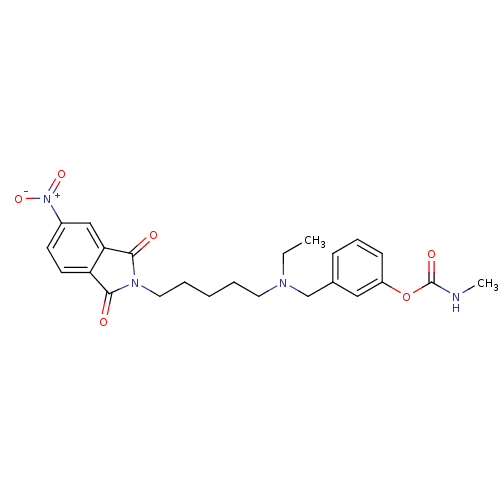 (CHEMBL106739 | Methyl-carbamic acid 3-({ethyl-[5-(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.380 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Chemical Industries, Ltd. Curated by ChEMBL | Assay Description Inhibition concentration against rat acetylcholinesterase | J Med Chem 37: 3141-53 (1994) BindingDB Entry DOI: 10.7270/Q2JS9PHX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM8963 (CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1016/j.bmcl.2022.128602 BindingDB Entry DOI: 10.7270/Q2HM5DF5 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50149212 (CHEMBL117521 | N-(1,2,3,4,8a,10a-Hexahydro-acridin...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Sunesis Pharmaceuticals Inc. Curated by ChEMBL | Assay Description Inhibitory concentration against Acetylcholinesterase in rat brain | J Med Chem 47: 3463-82 (2004) Article DOI: 10.1021/jm040031v BindingDB Entry DOI: 10.7270/Q2NC61ZJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM8963 (CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of rat cortex AChE using acetylthiocholine iodide as substrate measured after 8 mins in presence of BuChE inhibitor ethopropazine by Ellma... | Eur J Med Chem 132: 294-309 (2017) Article DOI: 10.1016/j.ejmech.2017.03.062 BindingDB Entry DOI: 10.7270/Q2HX1G33 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM8963 (CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute Curated by ChEMBL | Assay Description Inhibition of rat brain Acetylcholinesterase using acetylthiocholine as substrate in presence of BChE inhibitor ethopropazine by spectrophotometric m... | Bioorg Med Chem 27: 895-930 (2019) Article DOI: 10.1016/j.bmc.2019.01.025 BindingDB Entry DOI: 10.7270/Q2N87F3N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM9047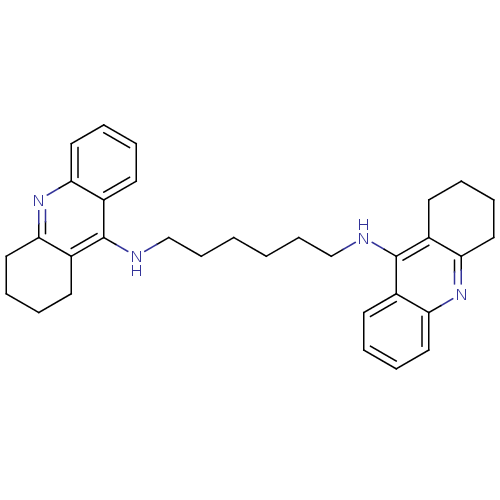 (Bis-THA inhibitor 5 | CHEMBL73800 | Hexylene-Linke...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of rat brain Acetylcholinesterase using acetylthiocholine as substrate in presence of BChE inhibitor ethopropazine | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00242 BindingDB Entry DOI: 10.7270/Q2KD22HB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50262878 (3-hydroxy-5-(3-(3-hydroxy-5-(trimethylammonio)phen...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Bari Curated by ChEMBL | Assay Description Inhibition of bovine AChE by Ellman's method | Bioorg Med Chem 16: 7450-6 (2008) Article DOI: 10.1016/j.bmc.2008.06.022 BindingDB Entry DOI: 10.7270/Q2NC611F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9024 (Indole-Tacrine Heterodimer 7 | N-[8-(6-Chloro-1,2,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50094626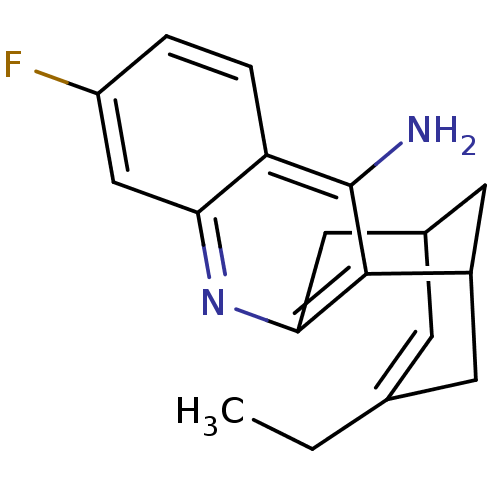 ((+)-15-ethyl-7-fluoro-10-azatetracyclo[11.3.1.02,1...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9038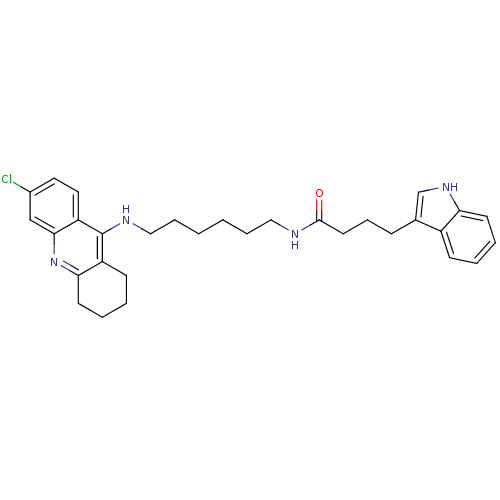 (Indole-Tacrine Heterodimer 21 | N-[6-(6-Chloro-1,2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM50398922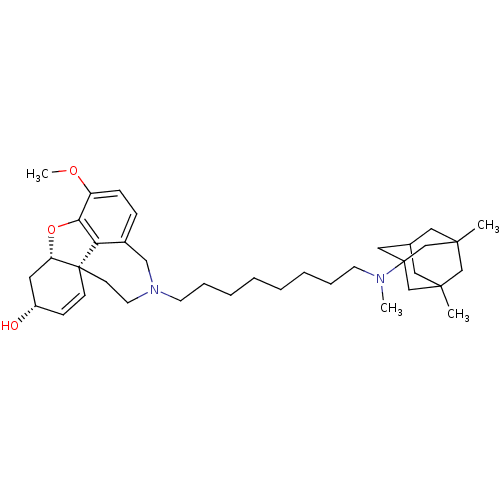 (CHEMBL2178790) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.520 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of AChE in Wistar rat brain homogenates using acetylthiocholine iodide and DTNB as substrate after 10 mins by Ellman method | J Med Chem 55: 9708-21 (2012) Article DOI: 10.1021/jm3009458 BindingDB Entry DOI: 10.7270/Q2VQ33TR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM9409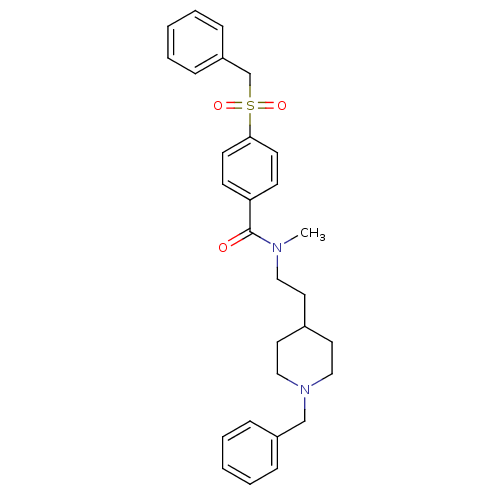 (CHEMBL299708 | N-[2-(1-benzylpiperidin-4-yl)ethyl]...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.560 | n/a | n/a | n/a | n/a | n/a | n/a |
Eisai Co., Ltd. Curated by ChEMBL | Assay Description In vitro inhibitory concentration against acetylcholinesterase (AChE) obtained from mouse brain homogenate. | J Med Chem 35: 4542-8 (1993) BindingDB Entry DOI: 10.7270/Q25M64P8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271141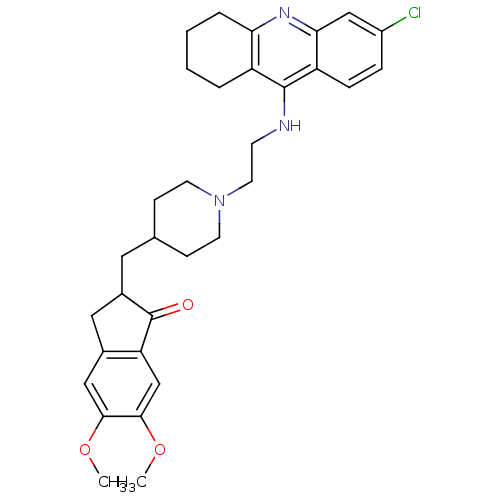 (6-Chloro-9-[(2-{4-[(5,6-dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM9051 (6-fluoro-N-{7-[(6-fluoro-1,2,3,4-tetrahydroacridin...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | 7.4 | 37 |
National Defense Medical Center | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. Enzyme activity was determined by measuring the absorbance at... | J Med Chem 45: 2277-82 (2002) Article DOI: 10.1021/jm010308g BindingDB Entry DOI: 10.7270/Q2KW5D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM9411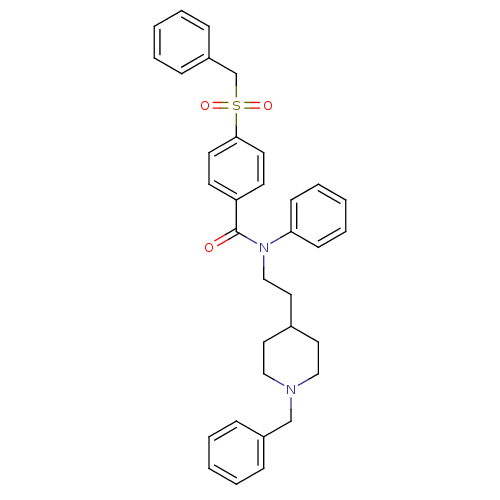 (CHEMBL54058 | N-[2-(1-benzylpiperidin-4-yl)ethyl]-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Missouri-St. Louis Curated by ChEMBL | Assay Description Inhibition against Acetylcholinesterase (AChE) | J Med Chem 39: 380-7 (1996) Article DOI: 10.1021/jm950704x BindingDB Entry DOI: 10.7270/Q25D8T1Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM9409 (CHEMBL299708 | N-[2-(1-benzylpiperidin-4-yl)ethyl]...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Missouri-St. Louis Curated by ChEMBL | Assay Description Inhibition against Acetylcholinesterase (AChE) | J Med Chem 39: 380-7 (1996) Article DOI: 10.1021/jm950704x BindingDB Entry DOI: 10.7270/Q25D8T1Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM9409 (CHEMBL299708 | N-[2-(1-benzylpiperidin-4-yl)ethyl]...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Eisai Company, Ltd. Curated by ChEMBL | Assay Description Inhibitory activity against acetylcholinesterase | J Med Chem 38: 4821-9 (1996) BindingDB Entry DOI: 10.7270/Q2QC045T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9036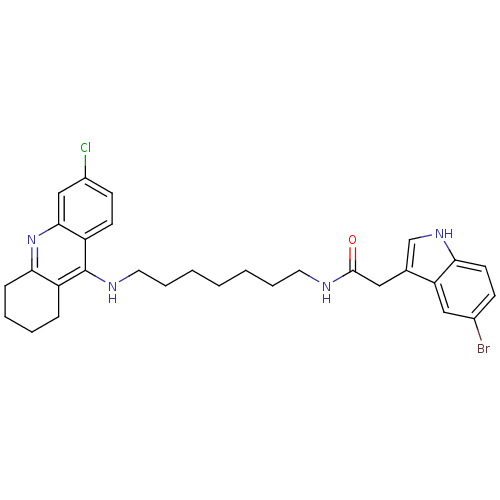 (2-(5-Bromo-1H-indol-3-yl)-N-[7-(6-chloro-1,2,3,4-t...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM9411 (CHEMBL54058 | N-[2-(1-benzylpiperidin-4-yl)ethyl]-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Laboratories | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The amount of a yellow substance formed after incubation was d... | J Med Chem 33: 1880-7 (1990) Article DOI: 10.1021/jm00169a008 BindingDB Entry DOI: 10.7270/Q20Z71H2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM9409 (CHEMBL299708 | N-[2-(1-benzylpiperidin-4-yl)ethyl]...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Laboratories | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The amount of a yellow substance formed after incubation was d... | J Med Chem 33: 1880-7 (1990) Article DOI: 10.1021/jm00169a008 BindingDB Entry DOI: 10.7270/Q20Z71H2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM9053 (6-chloro-N-{6-[(6-chloro-1,2,3,4-tetrahydroacridin...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | 7.4 | 37 |
National Defense Medical Center | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. Enzyme activity was determined by measuring the absorbance at... | J Med Chem 45: 2277-82 (2002) Article DOI: 10.1021/jm010308g BindingDB Entry DOI: 10.7270/Q2KW5D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Mus musculus (mouse)) | BDBM50379273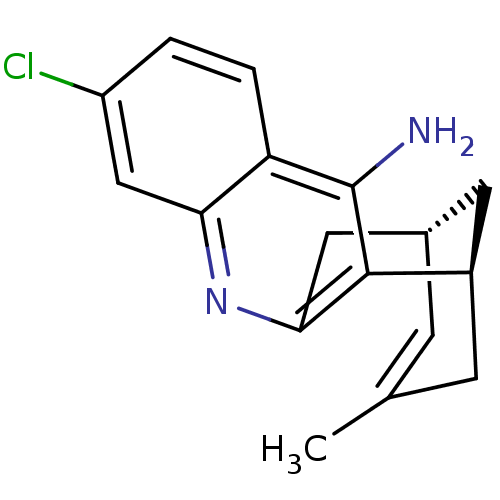 (CHEMBL1994202 | US9238626, (-)-Huprine Y HCl) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.620 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c02150 BindingDB Entry DOI: 10.7270/Q2F76HMM | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.640 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.640 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Rattus norvegicus (rat)) | BDBM8964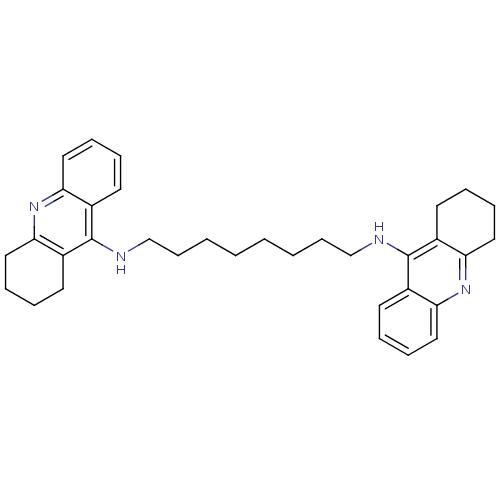 (CHEMBL75274 | Homodimeric Tacrine Analog 3c | N-[8...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.660 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute Curated by ChEMBL | Assay Description Inhibition of rat brain Acetylcholinesterase using acetylthiocholine as substrate in presence of BChE inhibitor ethopropazine by spectrophotometric m... | Bioorg Med Chem 27: 895-930 (2019) Article DOI: 10.1016/j.bmc.2019.01.025 BindingDB Entry DOI: 10.7270/Q2N87F3N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 3389 total ) | Next | Last >> |